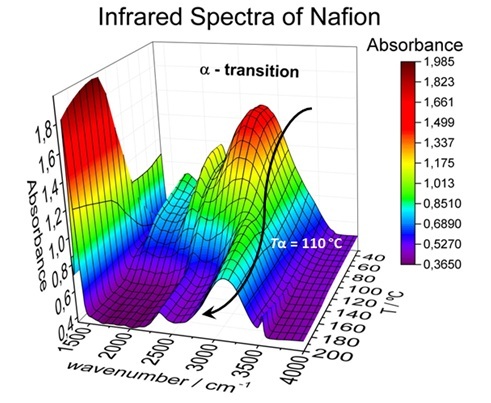
Nafion® is the most used polymer in Proton Exchange Membrane Fuel Cells (PEMFC) that work in the temperature range of 80-130 ºC. The PEMFC efficiency is controlled by a Nafion’s thermal transition, labeled α (Tα ~ 110 ºC). Above the α-transition, irreversible modifications of the Nafion chemical interactions is responsible for a dramatic degradation of Nafion proton conductivity that can lead to the fuel cell collapse. Such transition has been rarely studied by infrared spectroscopy (IR). The IR would allow understanding the specific interactions among Nafion’s functional groups underlying α-transition. Thus, this investigation would help boosting PEMFC technology as well as advancing the development high-performance polymer materials.
______________________________________________________________________________
The autor:

Jaqueline
HZB summer student
Universidade de São Paulo
Brasil

This is not exactly a catchy headline nor a picture which could attract interest of non experts, I had some difficulty to understand what is the issue. But for experts it might be very useful of course.
Interesting topic.
very interesting, and connected to my project …. i would like to hear more about it
Interesting subject. I would like to know more.