
Quantum Mechanics Can Be Useful Too!
Nowadays, it seems that quantum mechanics is seen more as “spooky” than practical. Thus, I am here to “de-mystify” some of the science behind a fascinating process that’s yet to be simulated properly. My research group investigates the theory behind what is called Interatomic Coulomb Electron Capture (ICEC). This fascinating universal quantum process is yet to be fully understood, and we are here to change that. Someday, this could be an incredibly useful tool to revolutionize atomic and quantum chemical energy transfers.
ICEC In A Nutshell
The general process includes 3 main steps that occur simultaneously. We’ve got two atoms, atom A and atom B (like in the diagram below), and we have a free electron. First, this free electron (with some energy ε) is shot in and is then captured into atom A’s orbit, which results in an emission of energy in the form of a photon. This energy is then absorbed by its neighboring atom B. This results in atom B ejecting an electron out of its orbit , with some energy ε’. This is what is called photo-ionization.
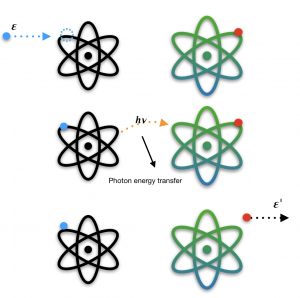
Why This Is Can Be Useful
This process can potentially change the way we see energy transfer. This is because the energy of the outgoing electron (with energy ε’ in Figure 1.) can be greater than the energy of the incoming free electron. This means that if we were to consider this process as an isolated system, it’s actually a very efficient way of “getting more than you put in”.
A Deeper Look Into The Interatomic Process
Let’s now think of this system, but a little more in depth about how each event transpires.
First, we throw in an electron in the direction of atom A. For our purposes, let’s ignore how exactly its being thrown in, just know that it is physically possible. From here, we have 2 main possible outcomes: either it can be scattered, which means that it bounces off the atom at some trajectory — or, it can be captured. Electron capture means that the electron is now in this atom’s orbit and it’s now part of the atom. Then comes the ionization/excitation!
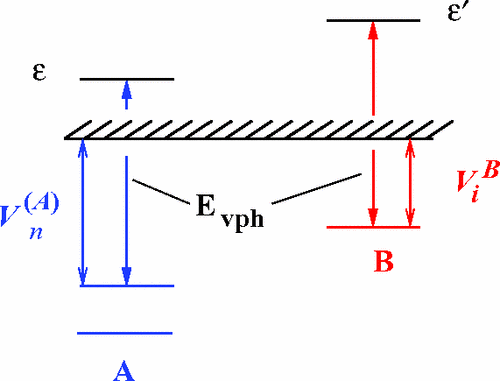
If you want to think of this thermally, the free electron shot in is like a hot bullet — in order to hold it, it has to be cooled down. That is why A passes of the energy. It literally heats up an electron on atom B in order to have it’s own electron cooler and bearably bound.
The Simulation Process
Our main task as scientists is to investigate the propability of this process occurring, given different sets of parameters. The probability of ICEC occurring depends on several different factors. It depends on the atomic species chosen, the distance between the two atoms, the energy at which the electron is thrown in at, and most importantly — the probability cross sections, which is where my focus lies.
A cross section is like the size of a target in archery or darts. How much “wiggle-room” do I have? I.e. How big is the target? Of course, the bigger the target, the better chances you have of hitting the target. And that’s kind of what a probability cross section does.
On a more technical side, in order to simulate such a process we use what is called Hartree-Fock method of computation. This is a developed way of approximating our system so that we can determine the wave function and energy of a many-body quantum system in a stationary state. This allows physicists like us to take this complicated family of probability distributions, and make it into something more tangible.
My goal is to create a framework in which we can predict the outcome of different scenarios and their probability cross sections. From this simulation, we can gather insights that wouldn’t have been possible otherwise. Even though we know it can observed, it doesn’t mean that we have any insight about the process itself. For instance, if you take the temperature of your city for a week and then plot it, does that mean you understand why the weather changed? Of course not. Hence, our drive to simulate ICEC. Our goal is to be able to fully understand the inner working of the process and what makes it so unique and universal. Then hopefully, we find some insight that can further our use of quantum energy transfers, and use it to our advantage.
What Is On The Horizon
As is the case with most theoretical physics, it can be very hard to predict the practical use of your research. Nevertheless, the most fruitful and important discoveries happen without an end goal in mind. Without these pursuits, we wouldn’t have the technology we are able to have today. Who would have known that a simple radio wave detection could leave to such incredible advances in technology. It’s only once we are able to understand how universal processes work that we can begin to postulate it’s role in society, and how it can contribute the advancement of knowledge and scientific discovery.
Citations:
[1] Gokhberg, K. & Cederbaum, L. S. (2010). Interatomic Coulombic electron capture. Phys. Rev. A, 82, 052707.

This sounds so interesting!
Yeah this was a good read. Thanks for sharing
Thank you for making quantum physics less scary for the rest of us 🙂
Energy capturing, wow, fascinating! Wonder where this will lead!
Wow! Very understandable from a non STEM background!
Thanks! I tried to give an explanation that would reach out to different scientific backgrounds, but I’m really happy to hear that I could provide a clear explanation even if you’re not in STEM 🙂
neat!
Nicely written and structured article. I like how you create a picture in the head of the reader which makes it easy to understand the process.
My comments to the physics:
The energy of the outgoing electron can be at most that of the incoming electron, which is due to energy conservation.
You might want to mention that the energy transfer is only possible due to the long-range Coulomb interaction between the two electrons on the different atoms. Otherwise, the excess energy would rather be emitted as a photon. (Since radiative decay is a much slower, ICEC is more likely to happen.)
It might be also helpful to understand the difference to the Auger process (i.e. why can the energy not be transfered to another electron in atom A).
The electron capture can also be followed by the ICD process, i.e. the electron in atom A relaxes to a lower bound state and the energy that is released there is handed to the electron in atom B. If I remember correctly, this sequence of processes is even more efficient.
I want to withdraw my comment to the energies: I forgot about the potential energy of the incoming electron. Thus, it is indeed possible that the outgoing electron can have a higher kinetic energy than the incoming one. Sorry about that 😉
Really nice and easy to understand explanation of the ICEC process. Very interesting project.
Thank you for the nice explanation. Though my field is totally irrelevant, thanks to your simple to understand explanation I feel I can grasp the concept.
A very good intro and effective explanation of a complicated phenomenon! A couple of question come to my mind:
Are you using the actual data from the observations in the simulation process?
Also, which elements are used as a target of the incoming electron?
And how does reactivity of the atom in the target element (Atom A) impact the result? (I assume this will be investigated using the simulation)
Looking forward to hear about the progress, best of luck!
Ni Nassi, so ICEC is known to be a phenomena that occurs, however it has not been experimentally observed. But there is analytical proof that it is theoretically possible. (However, a similar process called ICD, which doesn’t involve electron capture has indeed been experimentally observed). As of now we postulate to use heavier atoms such as rubidium and Barium(2+).
The probability of ICEC occurs does indeed has a lot to do with the reactivity of our atom A, but we shall see in the results to come some more insights regarding that!
This was an easy to follow explanation of what seems like a complicated and interesting topic. Best of luck with the simulations on it!
do you know if ICEC has been observed experimentally?
Hi Edward, so although ICEC has not been experimentally observed, we do have an analytical proof that it is theoretically possible. However, a similar process called ICD, which doesn’t involve electron capture component, has indeed been experimentally observed. Nonetheless, we can take it to be fact that ICEC inherently occurs, we just haven’t observed it yet per se 🙂
Thanks for the very clear explanation
Very good summary! ICEC is the most interesting process going on at HZB.
I agree!